The occupation numbers are now allowed to be, for instance, 0.9. 
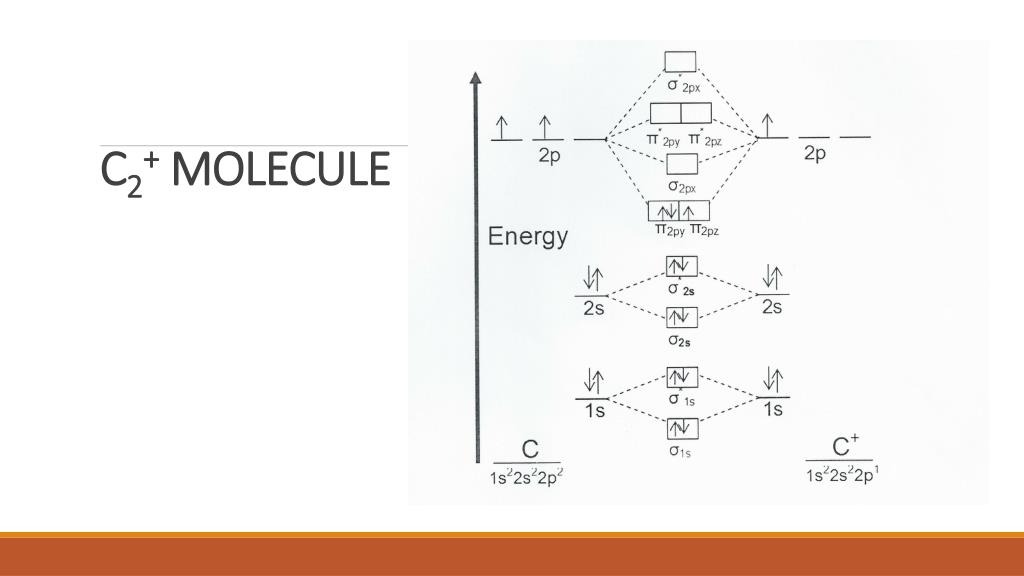
These new orbitals are called Natural Orbitals. The price paid to be able to calculate accurately binding energies is that we end up with orbitals with occupation numbers different from 1 (occupied) or 0 (unoccupied). When electron correlation effects are included, the “one-electron” in “one-orbital” picture is lost. However, the remaining 1% (called correlation energy, due to electron-correlation effects) is crucial in calculating binding energies within chemical accuracy. One may think that this is an astonishing achievement. With these “simple” models, one calculates roughly 99% of the total energy of a molecule. Obviously, the shape of the molecular orbitals resulting from the overlap of these orbitals is different. While VB theory allows for atomic orbital hybridization (leading to the known sp, sp 2, sp 3 hybridizations), MO theory assumes that no hybridization occurs at atomic level. The shape of the orbitals used in these approaches is one of the main difference between VB and MO. Earlier developed VB and MO methods where based on the model of each electron occupying an orbital. For molecules with more electrons, the exact solution is unaffordable in general, and approximate methods must be used. In fact, the exact total energies cannot be calculated for molecules with more than one electron, like H 2 +. Therefore, from the theoretical perspective, in order to calculate accurately the binding energy, we have to calculate accurately both the total energies at these two points. This value can be measured experimentally, and theoretical predictions must agree with the experimental value. Potential energy curve for a diatomic molecule.īinding energy, in diatomic molecules, is calculated as the energy difference between two geometries: the equilibrium geometry and the “infinitely” separated atoms (see Figure 1). Let us get into the details of the chemical bond of this intriguing molecule. In addition, they provide a connection with the apparently contradictory MO results. Our results predict a quadruple bond for this molecule, in agreement with recent VB calculations. 
In a recent publication 2, we have approached the problem using a different method, the Natural Orbital Functional theory (NOFT). Nevertheless, we have to keep in mind that both methods are approximate. Apparently, these two results are contradictory. While some calculations, based on Valence Bond (VB) theory predict a bond order of four 1 (quadruple bond) others, based on Molecular Orbital (MO) theory, predict a bond order of two (double bond). One may think that both are connected, but this is not always so, and, in the case of C 2, there is a vivid debate within the chemical community regarding it’s bond order. In this vein, two main aspects are clearly distinguished: i) the binding energy, or the energy gained by the molecule when two atoms are bonded and ii) the bond order (how many bonds are the responsible for such binding). One of the multiple tasks of quantum chemistry is the rationalization of the bonding between atoms to form molecules.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
